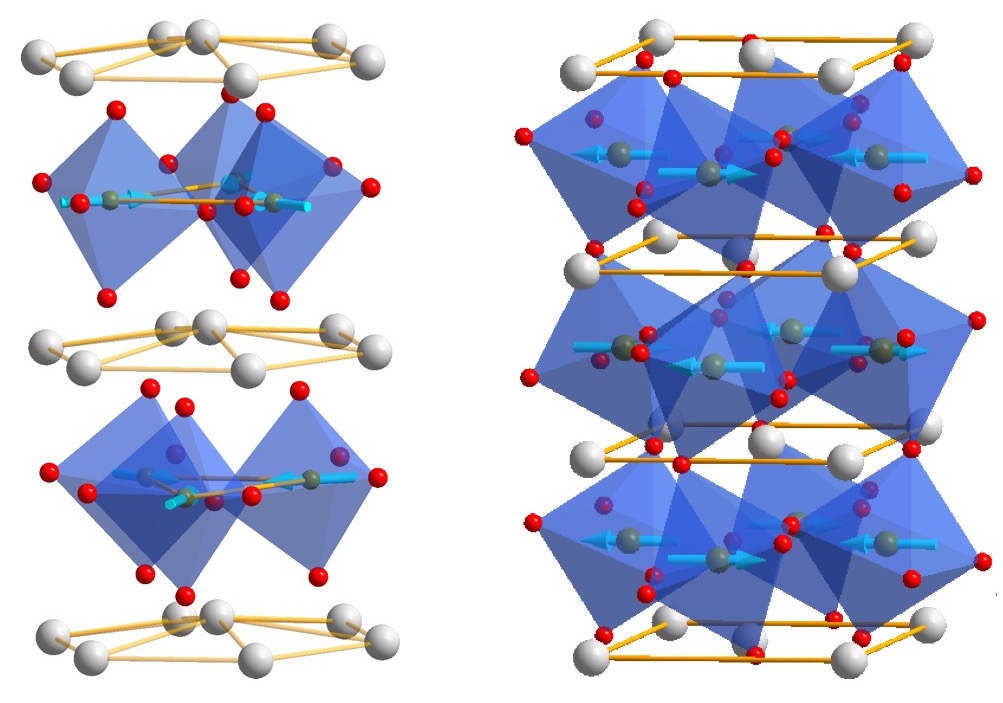
Grain Power!
xiaoshanxu2024-09-22T14:56:37-06:00Ferroelectric materials generate electric fields that move charges around, just as a bar magnet produces a magnetic field that moves magnets around. Ferroelectric materials can be used for data storage to make electronics more energy efficient, but they don’t always play well with the silicon technology used in devices like phones and computers. HAFNIA TO THE RESCUE! Click to learn more.