What would you like to search for?
The Shape of the Future
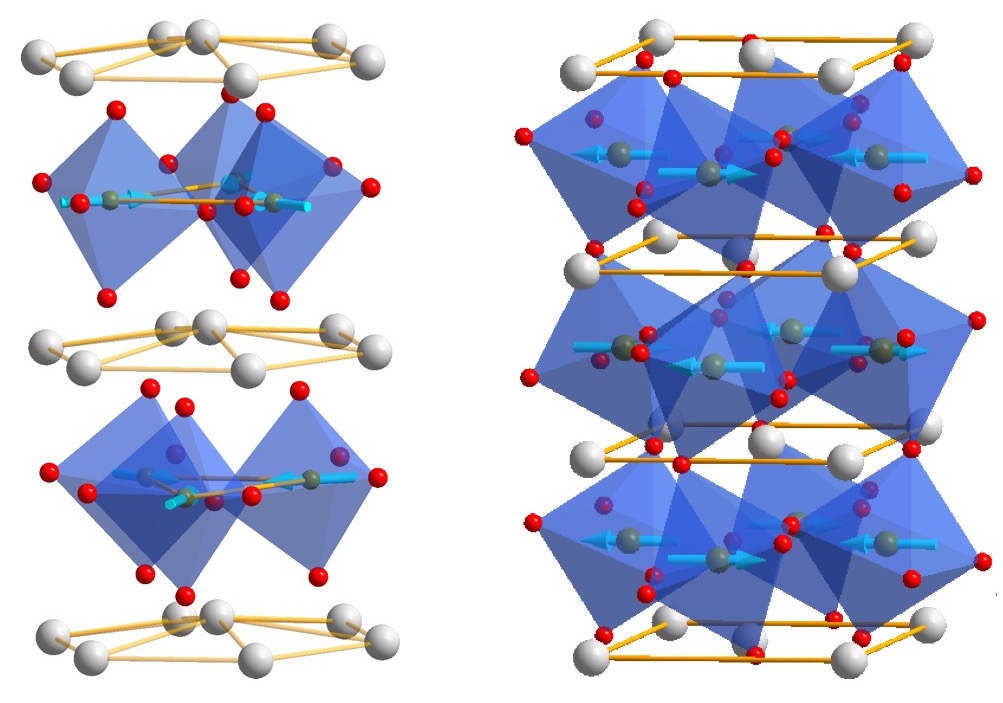
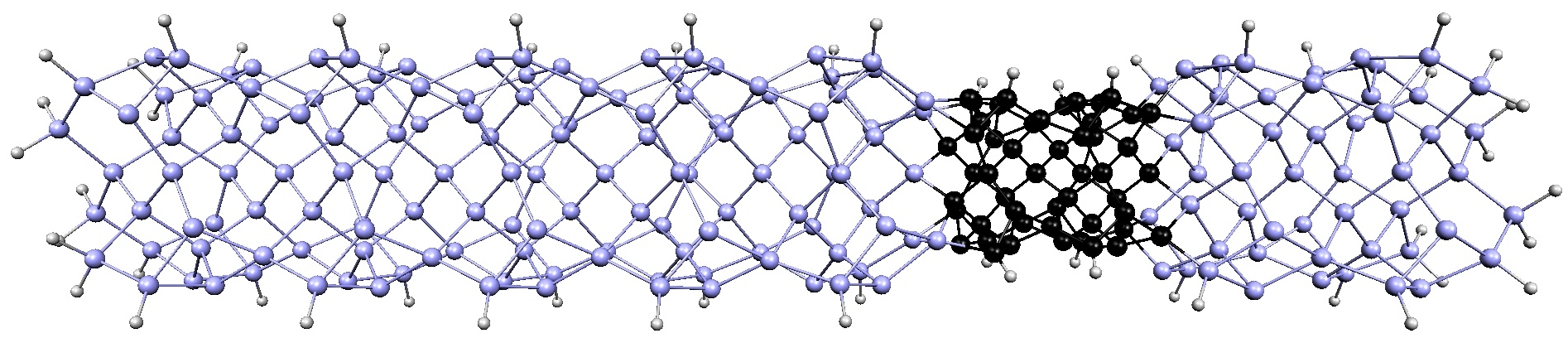
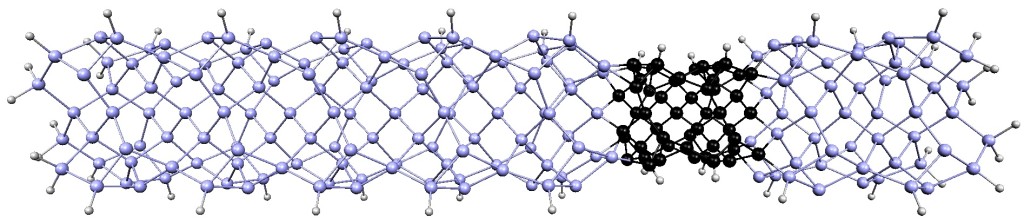
xiaoshanxu2021-07-14T11:04:27-06:00There are many ways atoms can arrange microscopically to form crystalline materials. Interestingly, materials created from different arrangements of the same atoms may exhibit completely different physical and chemical properties. A method called thin film epitaxy allows scientists not only to fine-tune the properties of known materials, but also to generate completely new materials with structures and properties not found in nature.


Interacting with the World’s Universal Building Blocks
WisconsinMRSEC2021-07-14T11:15:48-06:00Coloring INSIDE The Lines
wsliger2022-11-16T15:00:19-06:00Have you ever wondered why shining light on a glass of water causes rainbows to appear? Or noticed the colors that reflect from a CD or DVD? In this lesson, you will make an instrument called a spectroscope that can separate light into its hidden components. You will also be able to use the spectroscope to understand why different colored objects and light sources appear the way they do.


Superpowers of Liquid Crystals
WisconsinMRSEC2021-07-14T10:59:01-06:00It's a solid . . . it's a liquid . . . it's a LIQUID CRYSTAL! Researchers at the University of Wisconsin-Madison Materials Research Science and Engineering Center are investigating how the unique properties of liquid crystals allow them to act as environmental sensors, detecting toxins in the environment. In this video, we give a brief overview of what liquid crystals are and how their properties can be utilized to improve the world.


Make 21st-Century Wonder Material Graphene Cheaply and Easily in the Classroom!
WisconsinMRSEC2021-07-14T11:16:20-06:00Graphene is a two-dimensional material made from a single sheet of atoms, with outstanding mechanical, electronic, and thermal properties. It is a promising candidate to enable next-generation technologies in a wide range of fields, including electronics, energy, and medicine. This economical, safe, and simple lab activity allows students to make graphene via chemical vapor deposition in 30–45 minutes in a classroom setting.


Ferromagnetic liquids take shape
streubel2022-12-19T23:18:57-06:00You may have heard that there are three main phases of matter: solids, liquids, and gases (plus plasma if you want to get fancy). Liquids can take virtually any shape and deform instantly. Solid materials possess interesting electronic and magnetic properties essential to our daily life. But how about designing rigid liquids with magnetic properties? Impossible? Not anymore. Click to learn more!


How do Crystals get their Shapes?
funsizephysics2023-05-19T13:41:48-06:00Students make paper models of crystal unit cells and build a large crystal structure together while reflecting on the role of symmetry in crystal formation. This lesson is part 3 of a 4-part student-driven, lecture-free series, in which students will do card sorts, build hands-on models, solve engineering design puzzles, and more!


New World Disorder
Michael Randle2021-07-14T10:49:50-06:00We tend to think of materials as either electrical conductors or insulators: some materials, like metals, have low electrical resistance and conduct electricity easily, while others, like wood or plastic, have high electrical resistance and do not readily conduct electricity. Strange experimental results, however, reveal large fluctuations in the electrical resistance of thin metallic nanowires when a magnetic field or charge difference is applied to them. Click to learn how a more nuanced understanding of electron behavior helps to explain these variations in electrical resistance that may revolutionize the tech industry!


When you’re small, liquids behave like slippery solids
pelton2022-02-01T12:09:59-06:00We think we're pretty familiar with how ordinary liquids behave, but it turns out that some of the basic things we know are no longer true when we look at these liquids on short enough length scales and fast enough time scales. The liquids start to behave more like solids, pushing back when you push on them, and slipping across solid surfaces instead of being dragged along. Click to ride the tiny-but-mighty new wave of nanofluidics!


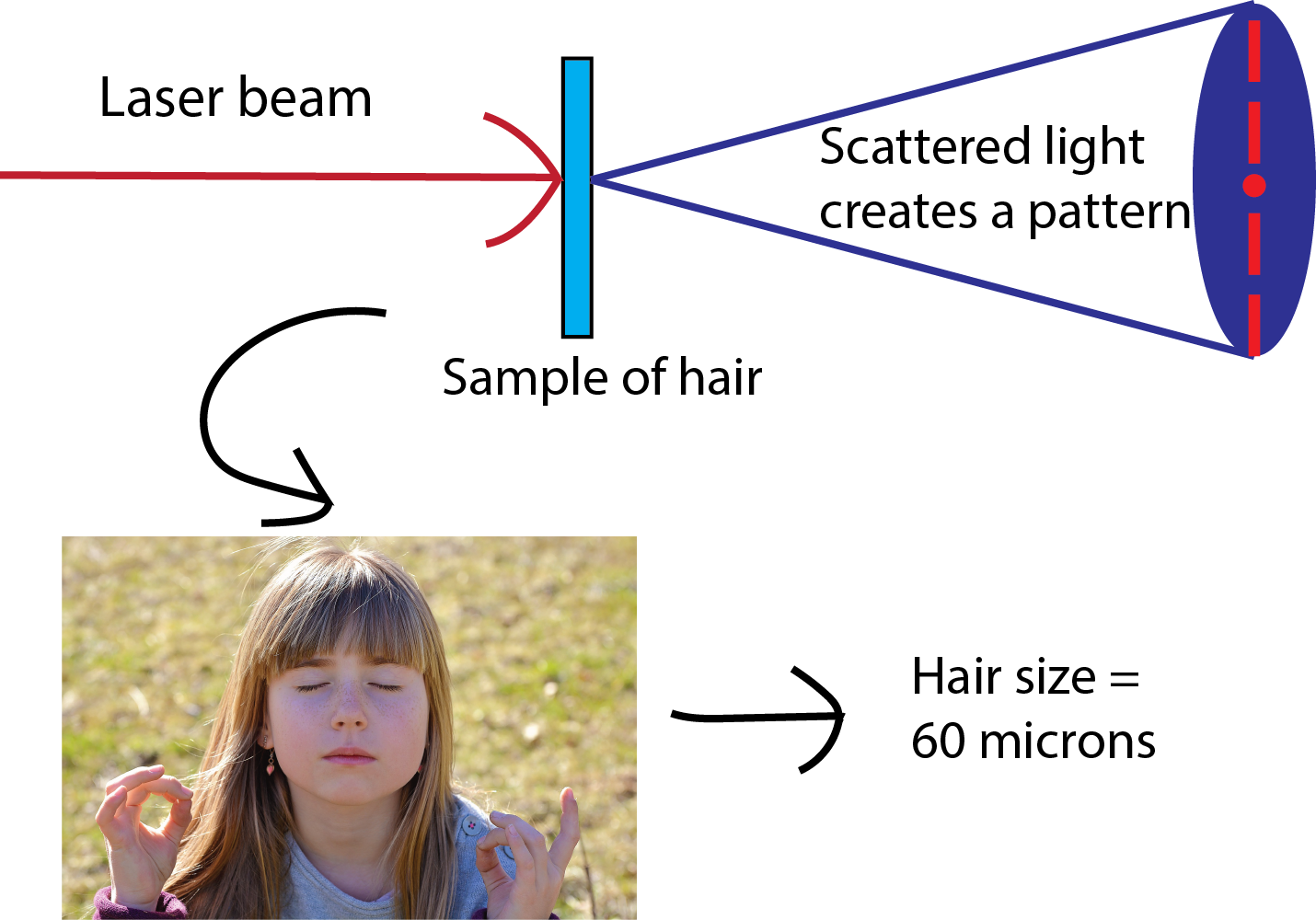
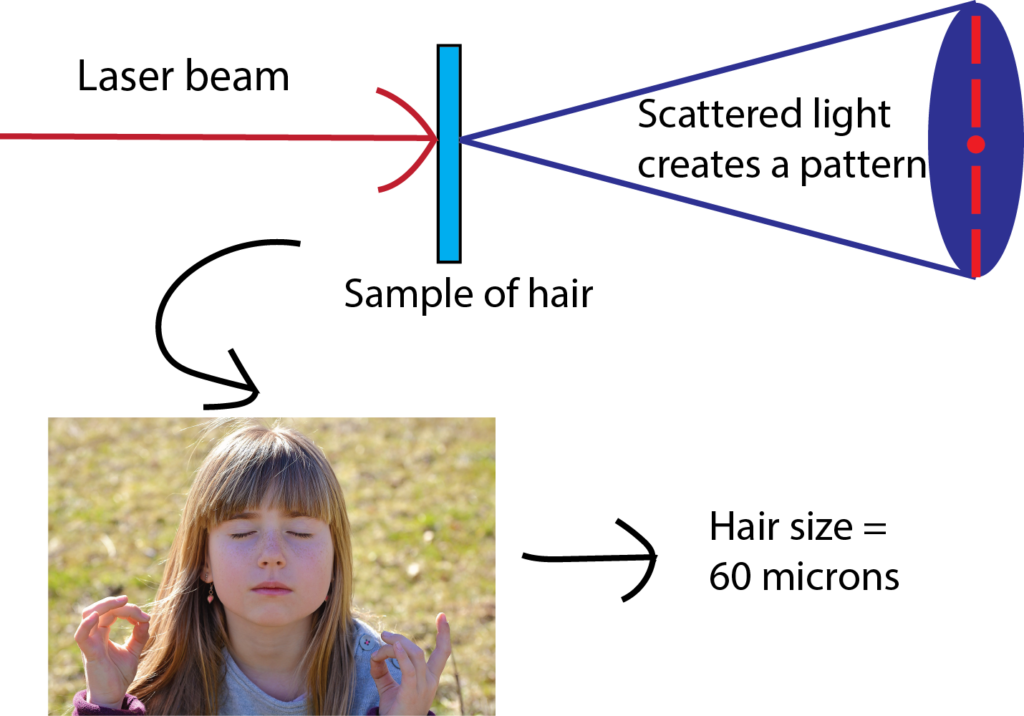
Use a laser pointer to measure the thickness of your hair!
bbrettmann2021-07-14T11:14:27-06:00Have you ever wondered how scientists can accurately measure the size of very small objects like molecules, nanoparticles, and parts of cells? Scientists are continually finding new ways to do this, and one powerful tool they use is light scattering. When an incoming beam of light hits an object, the light "scatters," or breaks into separate streams that form different patterns depending on the size of the object. This incoming light might be visible light, like the light we see from the sun, or it might be higher-energy light like X-rays. The light from commercial laser pointers, it turns out, is perfectly suited to measure the size of a human hair!


What is a Crystal, Anyway?
funsizephysics2023-05-19T15:33:43-06:00Crystals aren't magic, but they are amazing! In this engaging, comic-driven lesson, students do individual and group-based activities to understand the characteristics of crystals (like quartz) versus amorphous solids (like glass). This lesson is part 1 of a 4-part student-driven, lecture-free series in which students will do card sorts, build hands-on models, solve engineering design puzzles, and more!


Painting with magnets
sadenwalla12023-05-19T15:33:51-06:00Magnets curve themselves into beautiful patterns called domains, which cannot be seen with the naked eye. Now that magnetic paint and nail polish are easily available, we can use magnets to create all kinds of magnetic patterns which we can see, photograph, erase and rewrite! Click to find out how YOU can paint with magnets!


Dude, where my atoms at?
xiaoshanxu2021-07-20T11:06:51-06:00Have you ever wondered why some materials are hard and others soft, some conduct heat or electricity easily while others don't, some are transparent to light while others are opaque . . . and on and on and on? The material universe is vast and diverse, and while a material's properties depend in part on the elements it is made from, its structure—how it is built from its constituent atoms—can also have wide-ranging effects on how it looks, feels, and behaves. Diffraction is a method that allows us to "see" the atomic structure of materials. Read on to find out how it works!


Ketchup and oobleck and slime, oh my!
nicolesharp2021-04-21T14:08:43-06:00Why do so many fluids behave counterintuitively? Many substances in our lives – like oobleck, slime, or Silly Putty – don’t quite behave the way we expect a fluid to, despite some fluid-like properties. These substances fall into a special category: non-Newtonian fluids. Scientifically, this term is a bit of a catch-all for any substances that have a complicated relationship between their apparent viscosity and the force applied to them.


Superfluid helium and black holes
agdelma2021-07-14T10:53:26-06:00At low temperatures, helium—the same substance that makes balloons float—becomes a special type of liquid known as a superfluid, which has zero viscosity. It's like the anti-molasses! The properties of superfluids are governed by the laws of quantum mechanics. More specifically, the atoms in superfluid helium are “entangled” with each other, allowing them to share information and influence each other’s behavior in ways that are totally foreign to our everyday experience, and which Einstein famously described as "spooky action at a distance." Better still, scientists have recently discovered that the law controlling entanglement between different parts of a helium superfluid is the same as that governing the exotic behavior of black holes in outer space.


Carbon Onions
lukashev2021-07-14T11:02:46-06:00Carbon-based nanostructures are among the most intensely studied systems in nanotechnology. Potential practical applications span the fields of medicine, consumer electronics, and hydrogen storage, and they could even be used to develop a space elevator. A research team at the University of Northern Iowa is probing the properties of multilayered carbon nanostructures known as "carbon onions."


CHASING THE MYSTERIOUS AND ELUSIVE LIGHT HOLE
sophiahayes2021-07-14T10:29:14-06:00Semiconductors are materials with properties intermediate between metals and non-conducting insulators, defined by the amount of energy needed to make an electron conductive in the material. The non-conducting electrons occupy a continuum of energy states, but two of these states (the “heavy hole” and “light hole”) are nearly identical in energy. The heavy hole is easy to observe and study, but the light hole eludes most observers.


Gravity for photons
dwsnoke2021-07-14T10:33:51-06:00Inside solids, the properties of photons can be altered in ways that create a kind of "artificial gravity" that affects light. Researchers at the University of Pittsburgh tracked photons with a streak camera and found that whey they enter a solid-state structure, they act just like a ball being thrown in the air: they slow down as they move up, come to a momentary stop, and fall back the other way. Studying this "slow reflection" will allow us to manipulate light's behavior, including its speed and direction, with potential applications in telecommunications and quantum computing technologies.


Froot Loops, Legos, and Self-Assembly
aenders22021-07-14T11:15:04-06:00Self-assembly is the process by which individual building blocks—at the smallest level, atoms—spontaneously form larger structures. The structures they form depend on the size and shape of the building blocks, and on the conditions to which these building blocks are exposed. This can be demonstrated quite simply using breakfast cereal, or for more complex cases using specially prepared Legos.


How to Turn a Metal Into an Insulator
vandewal2021-07-14T10:30:43-06:00Solids are generally divided into metals, which conduct electricity, and insulators, which do not. Some oxides straddle this boundary, however: a material's structure and properties suggest it should be a metal, but it sometimes behaves as an insulator. Researchers at the University of California, Santa Barbara are digging into the mechanisms of this transformation and are aiming to harness it for use in novel electronic devices.


Heat Flow and Quantum Oscillators
stefanke2021-07-14T10:28:06-06:00Materials that are absolutely perfect—in other words, materials that contain no defect of any kind—are usually not very interesting. Imagine being married to a saint: you would quickly be bored out of your mind! Defects and impurities can considerably change many properties of materials in ways that allow a wide range of applications.


Swing-Dancing Electron Pairs
jeremylevy2021-07-14T10:44:45-06:00Superconductors are materials that permit electrical current to flow without energy loss. Their amazing properties form the basis for MRI (magnetic resonance imaging) devices and high-speed maglev trains, as well as emerging technologies such as quantum computers. At the heart of all superconductors is the bunching of electrons into pairs. Click the image to learn more about the "dancing" behavior of these electron pairs!


Use Light to Turn Your World Upside-Down!
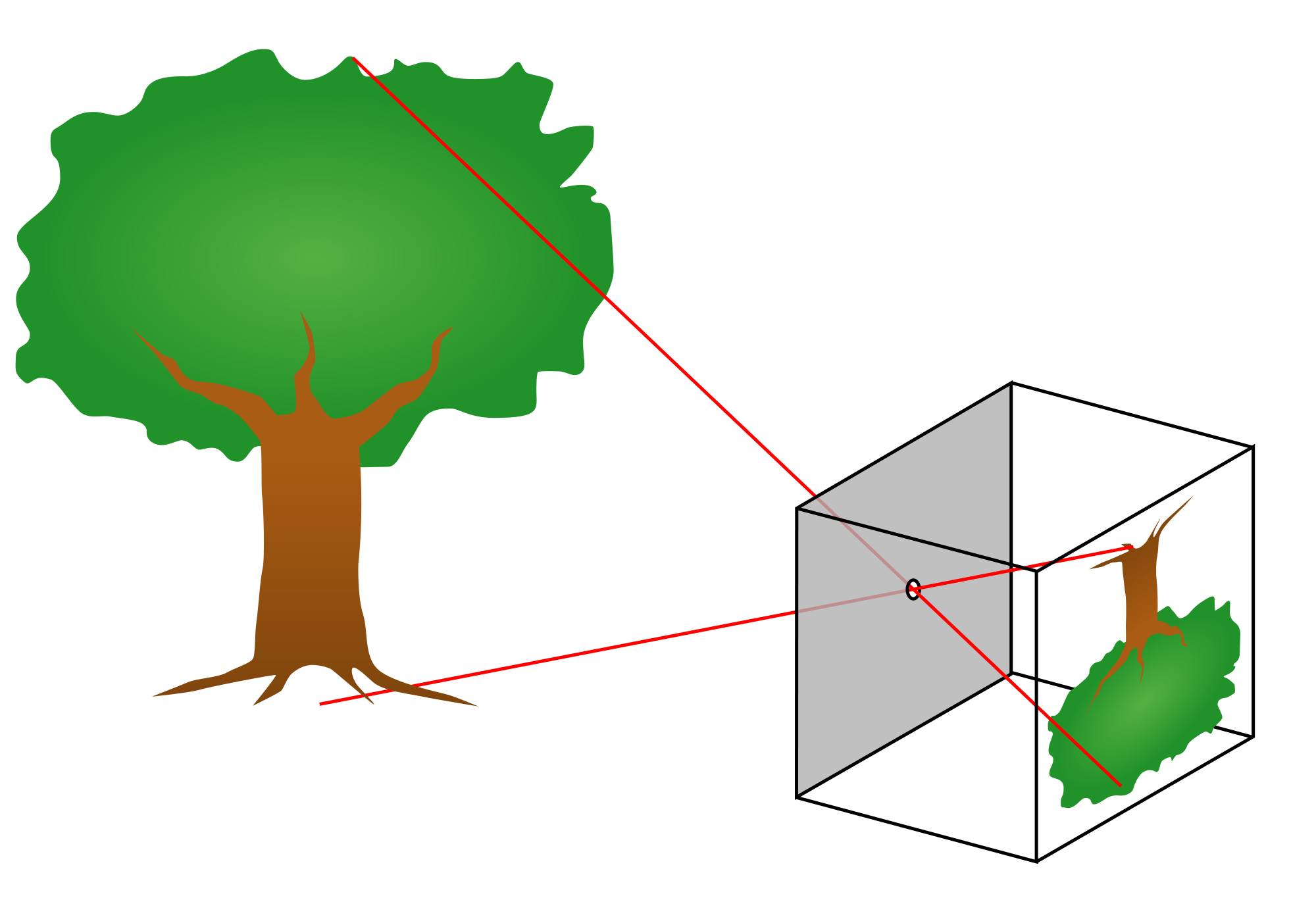
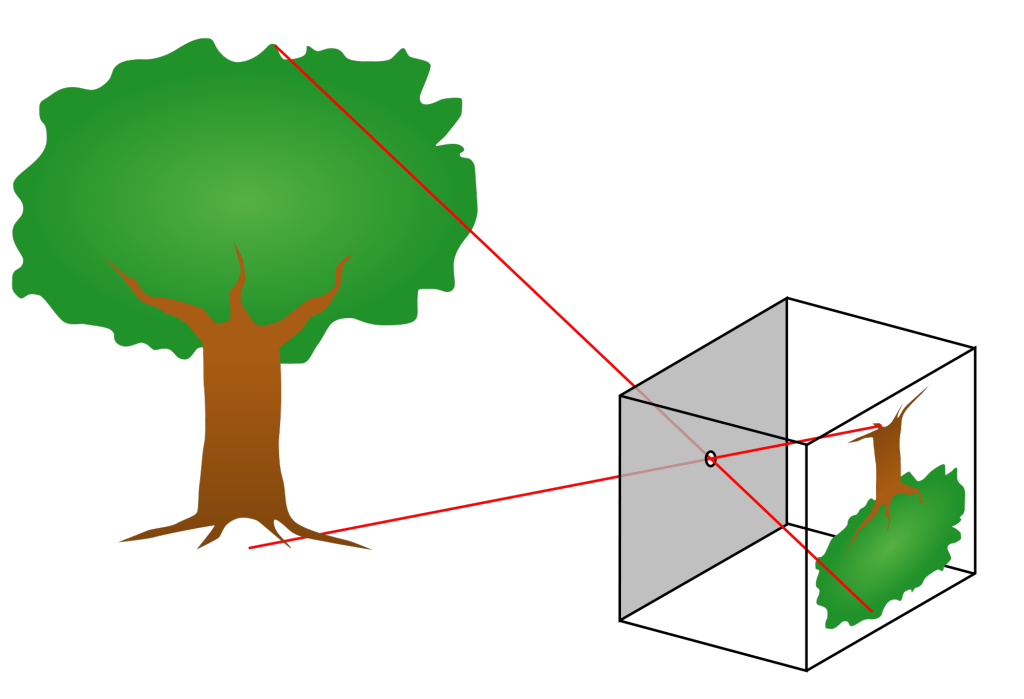
gvoth2021-07-14T11:17:45-06:00We can easily observe light with our eyes, and so it is one of the most familiar parts of the world around us. And yet, light often does amazing and unexpected things. Light travels in straight lines from the source to our eyes. This fact allows us to understand many of the cool things that light can do. In this lesson, we will observe how light creates mirages and shadows. And we will build a pinhole camera which makes things appear upside-down. We can understand the upside-down images by thinking about the straight line that the light took from the object to the screen.


Unit Cells and Their Molecular Building Blocks
funsizephysics2023-05-19T15:33:04-06:00Through hands-on activities using gumdrops and toothpicks, students will learn about unit cells that make up the repeating structures of crystals like table salt. This lesson is part 2 of a 4-part student-driven, lecture-free series, in which students will do card sorts, build hands-on models, solve engineering design puzzles, and more!